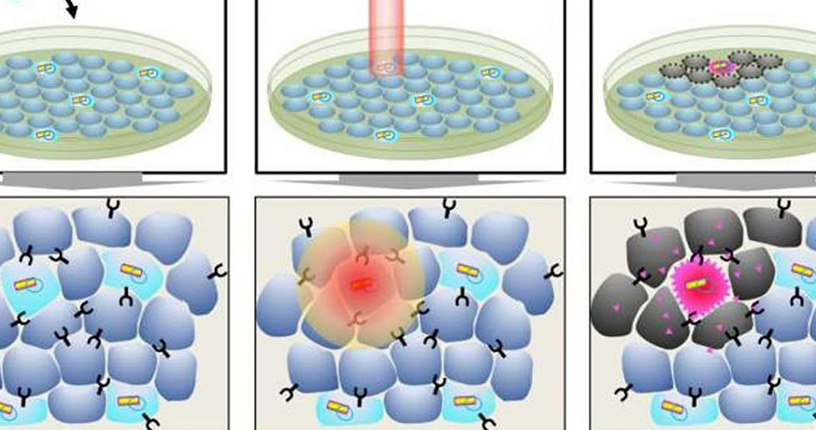
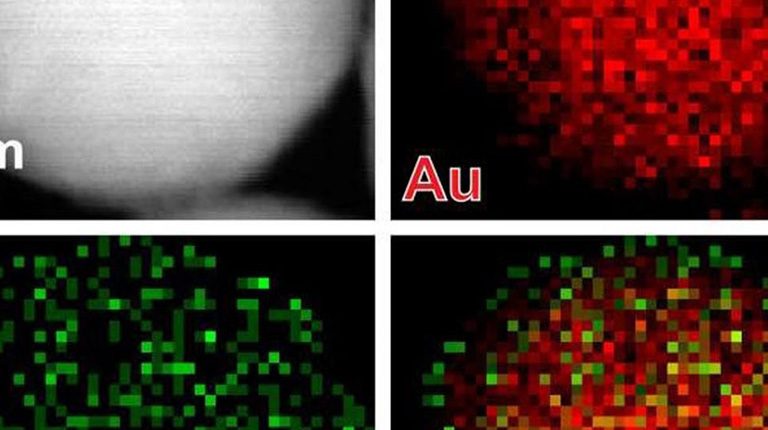
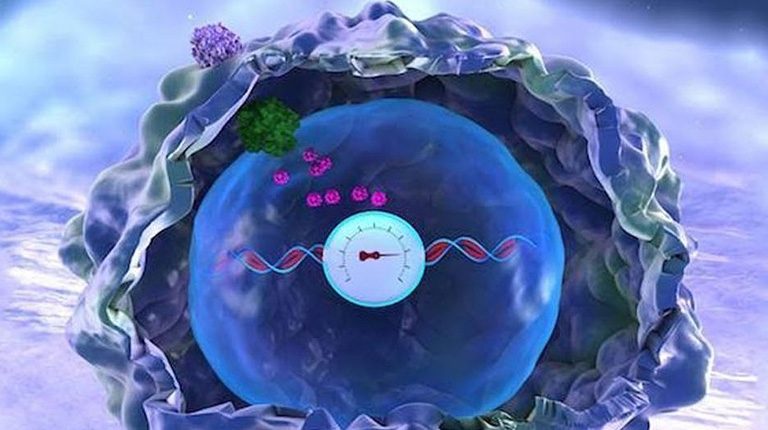
Researchers at the Institute of Cellular and Materials Science, Kyoto University of Japan, have invented a new method that, by correcting the level of nano-bars, facilitates the transfer of cancer-destroying genes into the cell in comparison with the past. This method involves the coating of gold nanoparticles and lipids that, when exposed to near infrared lasers, converge lipids into an oily liquid and increase the ability of nano-tubes to penetrate the cell.
The activation and release of the gene is carried out by nano-bars. Gold nanoparticles made functional with lipid to enhance the penetration of nanotubes into the cell.
Plasmid vector
The research team used plasmid carriers as gene carriers, which includes a
protein that reacts to heat. First, the carrier is bound to the increased green
fluorescent protein gene (EGFP) and is transmitted by the gold nanorods coated
with the lipid into the mammalian cells. The gene is illuminated. While
non-target cells lack the expression or expression of the EGFP gene, the gold
nanoscale carries a conductive protein called TRIAL that can penetrate into
cancer cells. In the next step, a protein called TRAIL is added to the plasmid
carrier. . This protein contains the necessary ingredients to destroy the
cancer cell line. Laser radiation into nano-tubes containing this protein
increases the death rate in cancer cells, which can potentially be useful in
the treatment of cancer molecules. This new system provides a new opportunity
for targeted targeting and accurate gene expression in mammalian cells by
minimizing laser imaging The results of this finding were published in the
journal Scientific Reports.
Source :
Hirotaka Nakatsuji et al, Surface chemistry for cytosolic gene delivery and photothermal transgene expression by gold nanorods, Scientific Reports (2017). DOI: 10.1038/s41598-017-04912-1